Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124

Food and Drug Administration (FDA) has approved the first test test at home for cervical crabThe product manufacturer posted on Friday.
I could serve the test as an alternative to a personal “Pap-Bris”, which are recommended every three years for women under 65 years.
The teal-stypic collection device, which made Teal Health in San Francisco-is prevented by women to get a sample and send it to a laboratory analysis.
The test is designed to detect human papilloma virus (HPV), a virus that causes almost all cervical cancer cases.
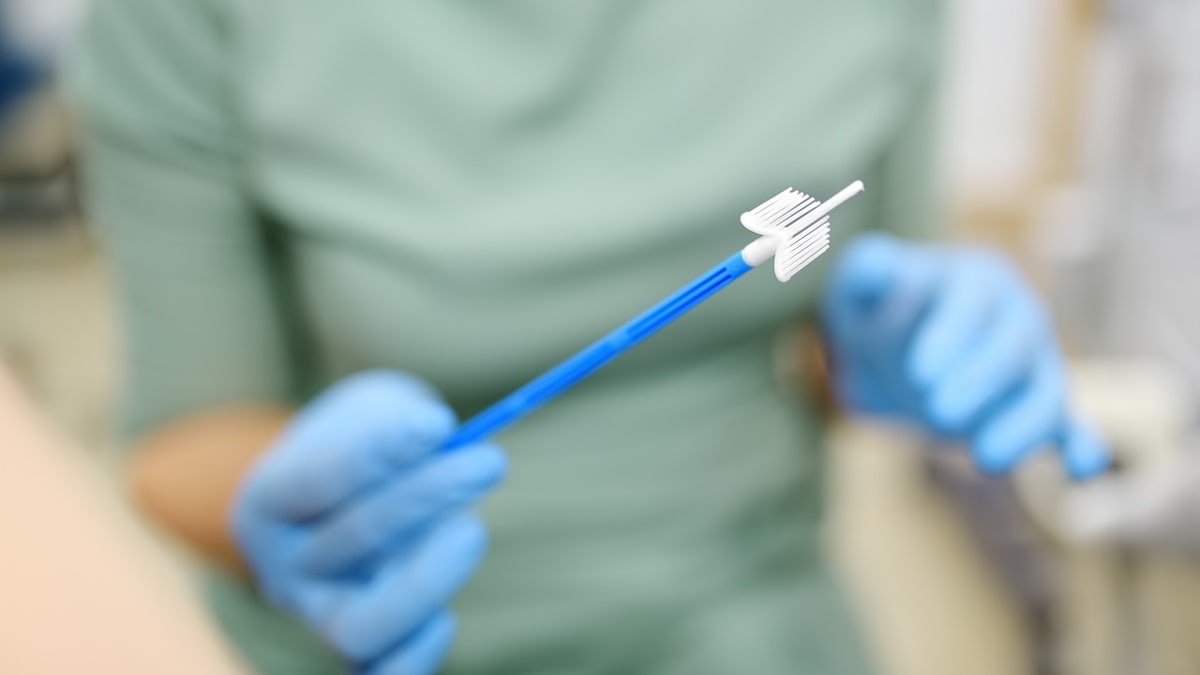
The food and medication administration approved the first test of screening at home for cervical cancer, the product manufacturer announced on Friday. (East)
Approval comes after a clinical trial, including More than 600 womenin which the Teal wand had 96% accuracy to detect the uterine precarcera.
In the study, 86% of participants said they were more likely to be in accordance with the recommendations for the burglary of cervical cancer if I could do so at home, said Teal Health.
In addition, 94% said they would rather collect independently at home until the results were accurate.
The death of a cervical cancer could be reduced by home HPV testing, they are located
“The uterine cervical cancer is largely prevented, but the rehearsal rate in the US is still lagging behind, and the approval of the FDA this device for self-speed at home is a critical step forward,” said Dr. Christine Conugeeski, an associate professor, Ob-Gyn and a director of a complex clinical trial in Colorado.
“It offers a way that is based on evidence to expand access without threatening accuracy,” she added in a statement.

“The cervical cancer can be prevented to large extent, but the rates of the now-are lags are lagging behind, and the FDA approval of this device for independently collecting sticks at home is a critical step forward,” the doctor said. (East)
Approximately 11,500 new ones Cervical cancer diagnosis They are made in the US every year, and the disease causes 4,000 annual deaths, according to centers for the control and prevention of the disease.
In addition to the risk of cervical cancer, and some more risky HPV cases may also cause others Types of canceraccording to experts.
“Any type of test that helps detect a cervical cancer is a victory.”
Women between the ages of 25 and 65 are exposed to average risk of cervical cancer will soon be able to order a test at home on www.getteal.comAccording to the company’s announcement.
Sets are expected to first become available in California starting from June, and it is planned to expand throughout the country as soon as possible, “the announcement states.
Click here to sign up for our Health Bulletin
“Teal cooperates with the main insurance providers and plans to have flexible payment options, helping to remove financial problems and ensure that more women have access to this preferred screening at home if they want it,” the company said.

About 11,500 new diagnosis of cervical cancer is made in the US every year, and the disease causes 4,000 annual deaths. (East)
In addition to collecting kits, the product also includes telecommunications with the support of medical providers throughout the procedure.
Women who tests HPV positively will refer to the traditional PAP spots. Those who do not test positively are tested are not considered the risk of cervical cancer and will not have to be re -examined for three to five years.
Click here to get the Fox News app
“Some women are afraid of the traditional swab of the PAP or consider the procedure to be unpleasant-as a result, they have put away this vital test,” said Ami Vaidya Dr. med. Gynecological oncology At the Hackensack University Medical Center, John Theurer Center, in a press release. (Not included in trial.)
For more health articles, visit www.foxnews.com/health
“This could be an important tool for a regular review of more women, especially those who do not have access to a doctor. Any type of test that helps detect detection cervical crab is a victory. “