Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
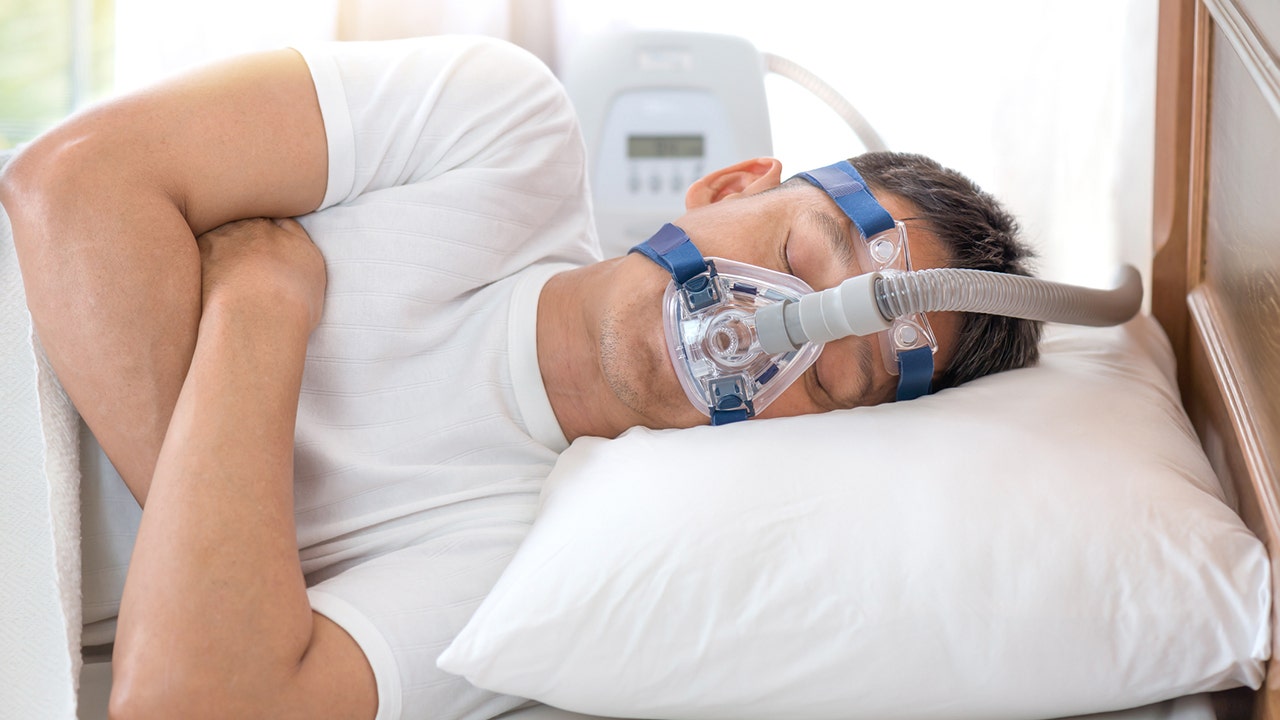
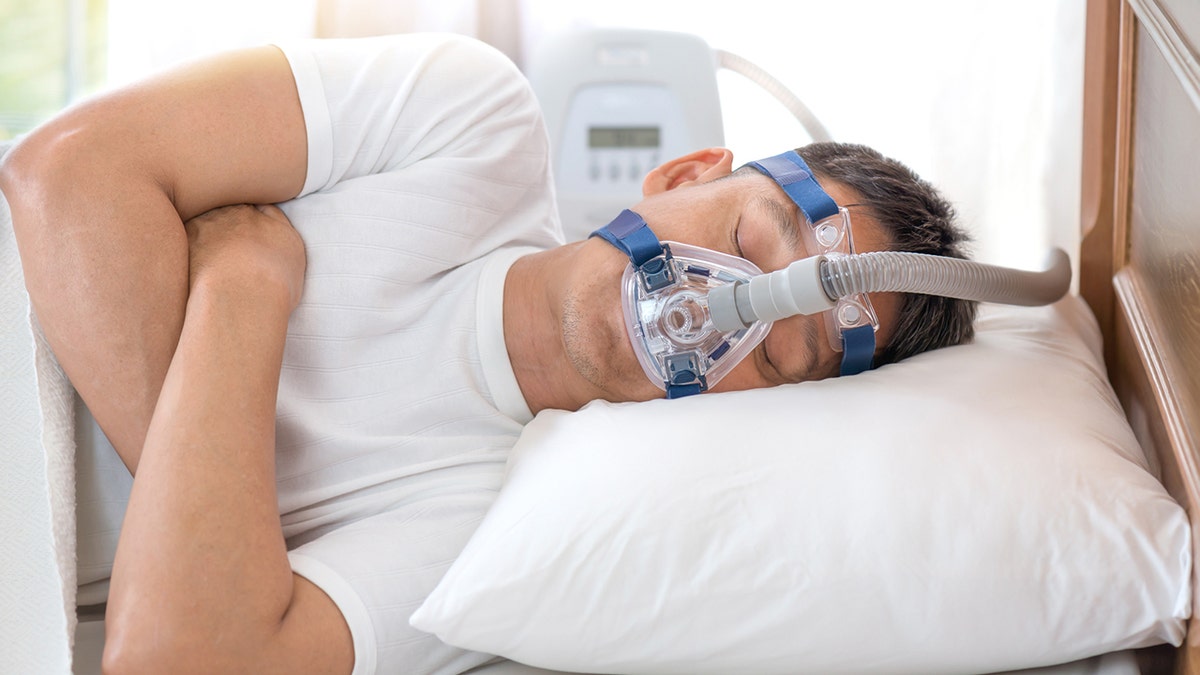
The first medicine for obstructive sleep apnea is approved by the US Food and Drug Administration (FDA).
On December 20, the FDA announced that the agency had approved Eli Lilly’s Zepbound (tirzepatide) for the treatment of moderate to severe obstructive sleep apnea (OSA) in adults with obesity.
The drug should be combined with a diet with reduced calorie intake and increased physical activity, the FDA pointed out.
SOME SLEEP APNEA PATIENTS SEE IMPROVEMENT WITH NEW BREATHING TOOL
Sally Seymour, MD, director of the Division of Pulmonology, Allergy and Critical Care at the FDA’s Center for Drug Evaluation and Research in Washington, DC, welcomed the approval in a release.
A happy and healthy elderly man sleeps deeply on his left side without snoring (iStock)
“Today’s approval marks the first drug treatment option for certain patients with obstructive sleep apnea,” Seymour wrote. “This is a major step forward for patients with obstructive sleep apnea.”
OSA occurs when the upper airway becomes blocked and causes pauses in breathing during sleep, according to the FDA. The condition is more common in people who are overweight or obese.
Similar to semaglutide treatments such as Ozempic and Wegovy, Zepbound activates gut-secreted hormone receptors (GLP-1 and GIP) at reduce appetite and food intake.
About 30 million people in the US suffer from sleep apnea
Studies show that by reducing body weight, Zepbound “also improves OSA,” the FDA noted.
In a 52-week study, participants treated with Zepbound experienced a “statistically significant and clinically meaningful reduction in apnea or hypopnea,” and a large proportion of participants achieved remission, or “symptom resolution.”

Studies show that Zepbound improves obstructive sleep apnea by reducing body weight. (iStock)
Patients treated with Zepbound also reported a significant reduction in body weight, the FDA mentioned.
The drug can reportedly cause side effects such as nausea, diarrhea, vomiting, constipation, stomach discomfort and pain, injection site reactions, fatigue, allergic reactions (usually fever and rash), belching, hair loss, and gastroesophageal reflux disease.
DO WOMEN NEED MORE SLEEP THAN ME? HERE’S WHAT THE EXPERTS THINK
Although Zepbound causes thyroid C-cell tumors in rats, it is not known whether it causes these tumors in humans, so it should not be used by patients with a personal or family history of medullary thyroid cancer or with multiple endocrine neoplasia type 2 syndrome, according to the FDA.
The agency encourages all OSA patients to consult a doctor before you take Zepbound and monitor for possible complications.

One sleep expert called the FDA’s approval “a promising advance for the millions of people who suffer from this condition.” (iStock)
Sleep expert dr. Wendy Troxel, RAND Corporation Senior Behavioral Specialist and Licentiate clinical psychologist in Utah, called the FDA’s approval “a promising advance for the millions of people who suffer from this condition.”
“Zepbound promotes weight loss and has been shown to reduce cases of apnea.”
About 30 million people in the U.S. suffer from sleep apnea, Troxel told Fox News Digital.
Although the most common treatment for sleep apnea – positive airway pressure (PAP) – is “very effective” in treating the condition, up to 50% of patients are “non-adherent”, she said.
CLICK HERE TO SUBSCRIBE FOR OUR HEALTH NEWSLETTER
“Zepbound promotes weight loss and has been shown to reduce the incidence of apnea,” noted Troxel.
“For obese individuals with moderate to severe sleep apnea, this new treatment option may offer an important alternative or adjunctive treatment, particularly for those struggling with sleep apnea therapies such as positive airway pressure.”

People with a history of a severe allergic reaction to tirzepatide should not use Zepbound, the FDA warned in a news release. (iStock)
Troxel added that treating sleep apnea “is critical not just for the patient health and well-beingbut also for their bed partners, who are often the ‘hidden victims’ of untreated sleep apnea, given that loud snoring and gasping are the primary symptoms,” she said.
For more health articles visit www.foxnews.com/health
dr. William Lu, medical director of Dreem Health in San Francisco, told Fox News Digital that obesity and sleep apnea are “two of the most common health conditions affecting Americans today.”
“We still need to emphasize changing the dietary and health habits of many, but this is a fantastic start.”
“And they go hand in hand,” he said. “For patients who are eligible and have no contraindications, tirzepatide has the opportunity to be a next-generation drug that can help people lose weight, reduce the severity of sleep apnea, and improve overall health.”
CLICK HERE TO DOWNLOAD THE FOX NEWS APP
“We still need to emphasize changing the dietary and health habits of many, but this is a fantastic start.”
Getting tested for sleep apnea “is critical to your health,” Lu said, and could be a requirement to get drug coverage.
Fox News Digital has reached out to Eli Lilly for comment.